 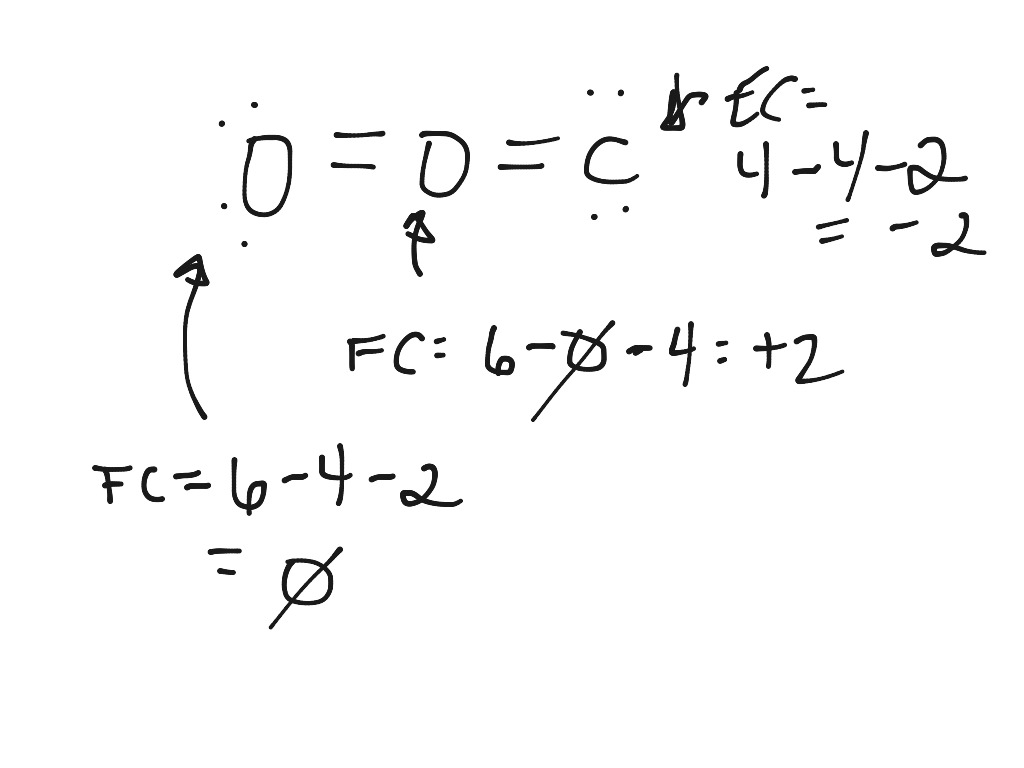
The structure is surrounded by brackets and has a superscripted negative sign. Assign formal charges to each atom in the interhalogen ion ICl4 ICl 4. Formal charge is only a useful bookkeeping procedure it does not indicate the presence of actual charges. The actual charge, however, is the actual electron density that is present on. We must remember that the formal charge calculated for an atom is not the actual charge of the atom in the molecule. Formal charge is a bookkeeping tool that is important to help us keep track of the electron flow in the reaction. Now, I’ve mentioned earlier that there’s a difference between the formal and the actual charge. The right structure shows a carbon atom with two lone electron pairs double bonded to a sulfur atom that is double bonded to a nitrogen atom with two lone electron pairs. The Difference Between the Formal and Actual Charge. Below this structure are the numbers negative two, positive one, and zero. The structure is surrounded by brackets and has a superscripted negative sign. The formal charge is a tool for estimating the distribution of electric charge within a molecule. The middle structure shows a carbon atom with two lone pairs of electrons double bonded to a nitrogen atom that is double bonded to a sulfur atom with two lone electron pairs. In order to calculate the formal charges for HNO3 we'll use the equation:Formal charge of valence electrons - nonbonding val electrons - bonding ele. Below this structure are the numbers negative one, zero, and zero. The structure is surrounded by brackets and has a superscripted negative sign. Formal chargeH 1 (1/2) × 2 0 0 This applies to each hydrogen. For formal charge, we act like each atom in a structure is an ion that has assigned to it all non-bonding electrons and half of all bonding electrons. Calculating the formal charges is important as it plays a crucial role in determining the minimum energy configuration among various possible Lewis structures. 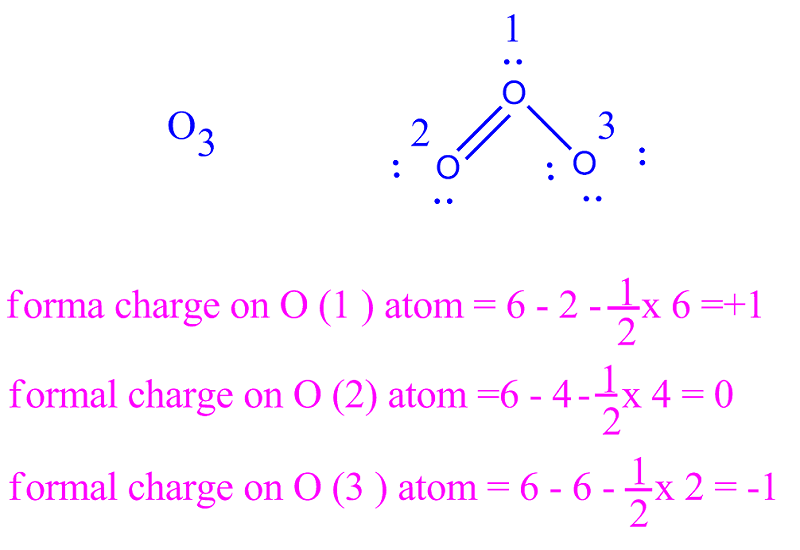
The top row is labeled, “Structure” and depicts three Lewis structures and the bottom row is labeled, “Formal charge.” The left structure shows a carbon atom double bonded to a nitrogen atom with two lone electron pairs on one side and double bonded to a sulfur atom with two lone electron pairs on the other. For calculating the formal charge, you need to remember this formula Formal charge Valence electrons - Nonbonding electrons - (Bonding electrons)/2. Formal charge is attempting to identify regions of high and low electron density within a structure, which help predict how some compounds react. Two rows of structures and numbers are shown. Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
